已传文件:photo/1772675767.png
Bone defect repair remains a major clinical challenge because autografts are limited by donor availability, and biologically inert implants usually require secondary surgery. Absorbable materials, including polymers, ceramics, and metals, are rapidly transforming orthopedic care by providing temporary support and synchronous degradation to match tissue regeneration. This review focuses on the cutting-edge advances in composite scaffolds, which enhance mechanical integrity, degradation kinetics, and osteoinductivity.We further explored innovative systems integrating stimulus-responsive smart materials (such as photothermal and piezoelectric materials), 4D-printed smart materials, regenerative therapies, and bone organoids. Emerging clinical data are discussed in the context of diseases such as osteonecrosis, osteomyelitis, and osteoporosis. Looking ahead, we propose transformative directions including artificial intelligence-driven scaffold design, gradient architectures, and immunomodulatory strategies to address the spatiotemporal mismatch between degradation and regeneration. This review provides a forward-looking framework for next-generation multifunctional, bioresponsive orthopedic implants.

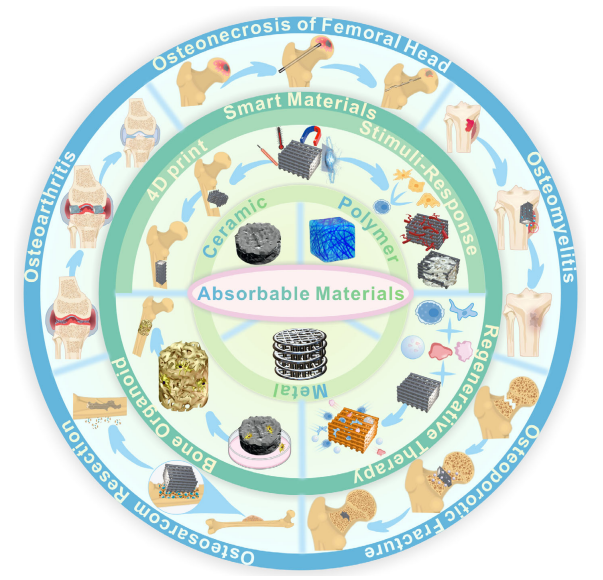
Construct orthopedic scaffolds using absorbable materials to achieve a dynamic balance between mechanical performance, degradation rate, and new bone regeneration.
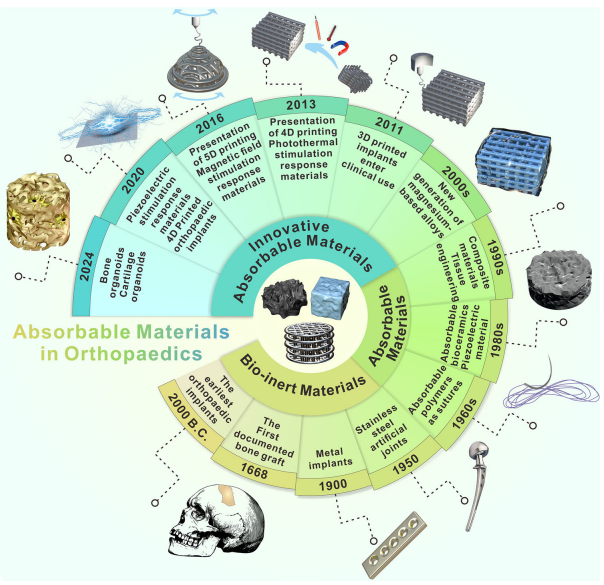
Development of absorbable scaffolds in orthopedics.
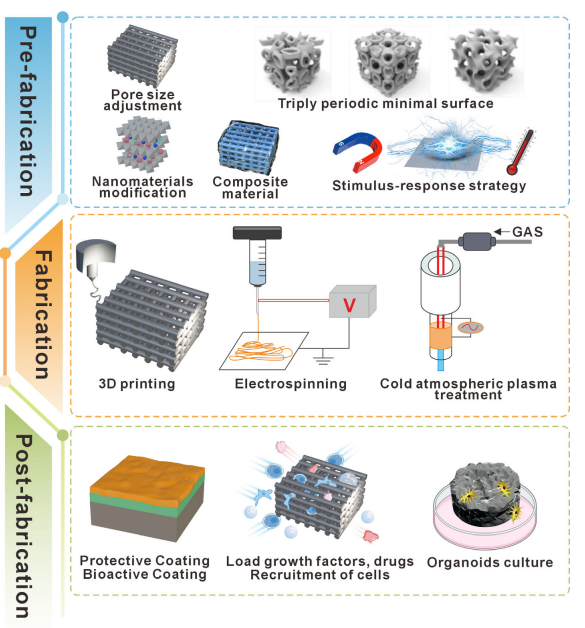
Common modification strategies for absorbable materials at different stages. In the prefabrication stage: structural design, surface modification, composite formulation, and stimulus-responsive engineering. In the manufacturing stage: plasma treatment and high-resolution 3D printing. In the post-processing stage: functional coatings for controlling degradation, osteogenesis, and inflammation regulation. Reproduced with permission [121]. Copyright 2022, Wiley-VCH
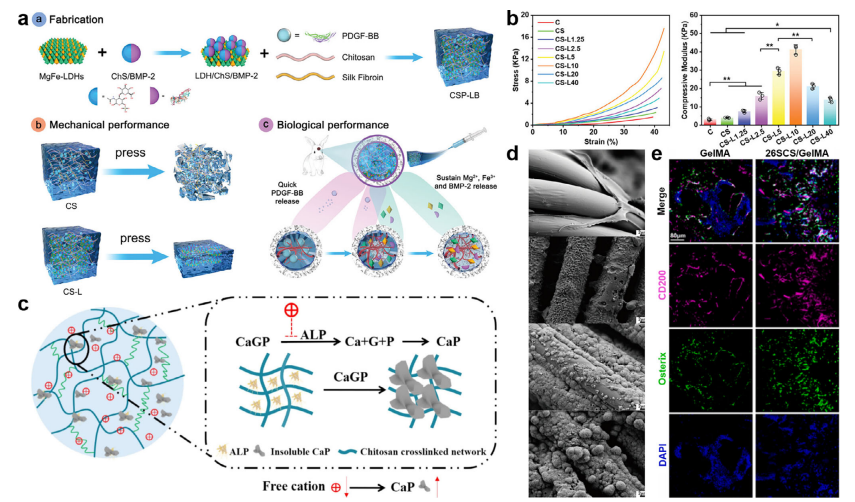
Chitosan-based absorbable materials for orthopedic applications. (a) Schematic diagram of CSP-LB hydrogel preparation and its functional mechanism. (b) Compression stress-strain curves (left) and compression modulus (right) of different samples. Reproduced with permission [173]. Copyright 2023, Wiley-VCH. (c) Schematic diagram of how the cationicity of chitosan hydrogel affects the mineralization yield.(d) SEM images of bone scaffolds before and after CAP treatment, as well as images after being soaked in SBF for 10 days. From top to bottom: pure CS, CS (CAP), CS/1.0 wt% HA, and CS/1.0 wt% HA (CAP). Reprinted with permission [176]. Copyright 2025, Elsevier. (e) Representative confocal images showing immunostaining of CD200 (purple) and Osterix (green), along with DAPI (blue) nuclear staining, in the femoral defect area of mice at 4 weeks post-surgery. Reprinted with permission [177]. Copyright 2021, Elsevier. Conductivity, antibacterial performance, hydrophilicity, and low immunogenicity [126, 170].
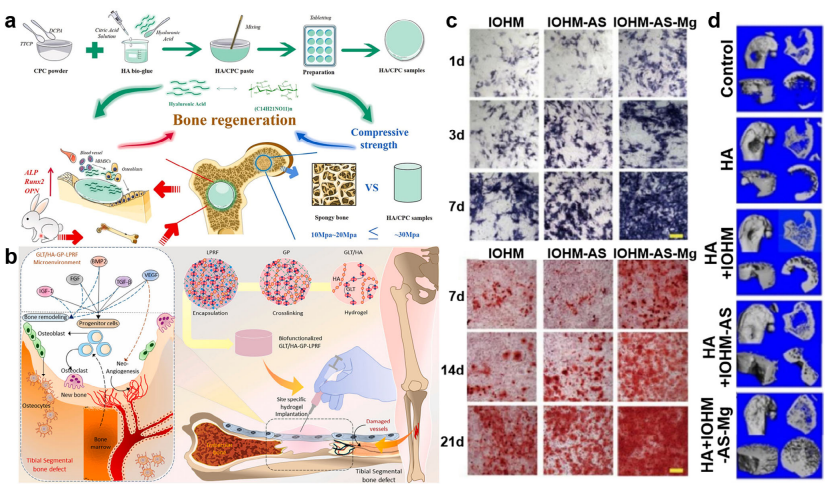
HA-based absorbable materials for orthopedic applications. (a) Schematic of HA/CPC composite material enhancing osteogenic gene expression and supporting bone repair. Reproduced with permission [181]. Copyright 2021, Elsevier. (b) Schematic of the mechanism of GLT/HA-GP-LPRF in treating segmental bone defects. Reproduced with permission [184]. Copyright 2024, Elsevier. (c) ALP staining (top) and ARS staining (bottom) of different hydrogels co-cultured with BMSCs in vitro. (d) Micro-CT images of femoral osteoporotic defects in different groups. Reproduced with permission [185]. Copyright 2024, Elsevier
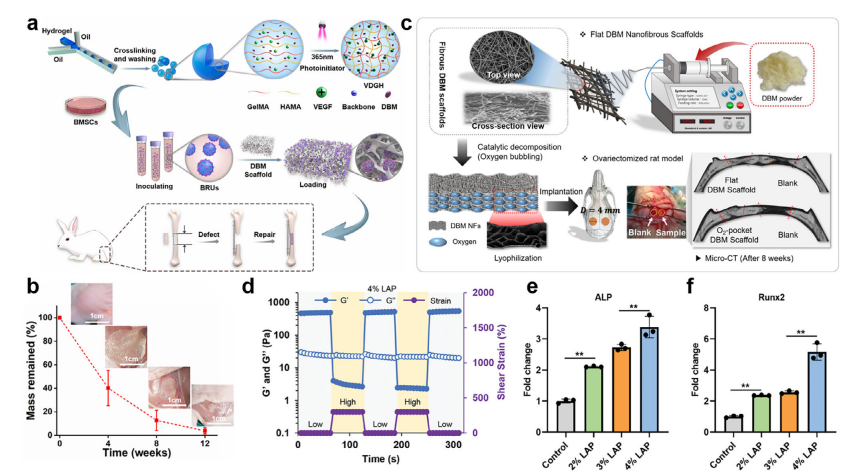
DBM-based absorbable materials for orthopedic applications. (a) Schematic diagram of repairing large-scale bone defects by combining a decalcified bone matrix framework with a photo-crosslinked osteogenic microgel bone regeneration system. (b) In vivo degradation assessment of VDGH microgels in the subcutaneous environment of nude mice. Reproduced with permission [190]. Copyright 2022, Elsevier. (c) Schematic diagram of electrospun DBM composite scaffold preparation and bone repair process. Reproduced with permission [191]. Copyright 2024, Elsevier. (d) Step-strain measurement of G-HA/4%LAP to characterize the self-healing ability of G-HA/LAP hydrogel. (e,f) Expression of osteogenic gene markers ALP and Runx2 evaluated on day 7 using qRT-PCR. Reproduced with permission [192]. Copyright 2024, Wiley-VCH. It has good biocompatibility and biodegradability, and contains abundant chemical sites, which are conducive to functionalization [131, 132].
Summary
Absorbable materials are reshaping the future of orthopedic treatment. This review systematically outlines the evolutionary path from traditional bioinert materials to absorbable polymers, metals, ceramics, and their composites, focusing on the key challenge of synchronizing degradation rate with new bone regeneration. In terms of material design, the research highlights how composite strategies are used to leverage advantages and offset weaknesses: absorbable metals like magnesium and zinc provide mechanical support, natural polymers such as chitosan and hyaluronic acid impart bioactivity, and ceramics like β-TCP and hydroxyapatite induce osteogenesis, ultimately constructing an intelligent scaffold that can bear load in the early stage while gradually degrading in tandem with bone tissue growth.
Looking further ahead, the article envisions innovative directions such as 4D-printed shape-memory scaffolds, photo-thermal/piezoelectric responsive materials, and bone-like organ construction. These technologies transform implants from static fillers into "living" materials that can actively deform to fit defects in response to body temperature or light, or generate electrical signals under mechanical stimulation to promote repair. From hip-preserving treatments for femoral head necrosis to antibacterial therapy for osteomyelitis and postoperative prevention of osteosarcoma recurrence, these materials have already shown potential in clinical applications. Although challenges remain in precise degradation control, large animal validation, and standardized evaluation, the deep integration of materials and regenerative medicine is driving orthopedic implants from passive replacement towards active repair.
References:
DOI: 10.1002/adma.202510848










